 This is also the featured periodic table on the cover of our Periodic Table Journal available at Amazon. The mole concept can be extended to masses of formula units and molecules as well. g/mol Check Convert the value 46. Enter a chemical formula to calculate its molar mass (e.g. Question: Using a Periodic Table with Two Decimal Places, calculate the Molar Mass of Sr(CIO), with correct Significant Figures. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in B2H6: Molar Mass (g/mol) B (Boron) 2 × 10.811 21.622. According to the periodic table, 1 mol of U has a mass of 238.03 g, so the mass of 2 mol is twice that, or 476.06 g. Periodic table of the elements Chemistry calculators Image gallery. It includes element names, symbols, groups, atomic numbers, and atomic masses. The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g. It features our favorite color scheme of all the tables we’ve made. 
In Chapter 1, we described Dalton’s theory that each chemical compound has a particular combination of atoms and that the ratios of the numbers of atoms of the elements present are usually small whole numbers. This 118 element periodic table is a 1920×1080 HD wallpaper. When calculating molecular weight of a chemical compound, it tells us how many grams are in one mole of that substance. Finding molar mass starts with units of grams per mole (g/mol). formula mass indicates that each nicotine molecule contains two formula units. Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the substance. To confirm your answer, compare the calculated value to the weighted mass displayed on the periodic table.\right ) \right ]=310.177\ amu \notag \) Atoms molar masses to convert the mass of each element to a number of moles. The atomic mass of oxygen (use percent natural abundance data from Table 2.5.1) would be calculated as follows:Ītomic mass = (%1)(mass 1) + (%2)(mass 2) + (%3)(mass 3) Periodic Table: Representative Elements & Transition Metals. For chlorine, there are only two naturally occurring isotopes so there are only two terms.Ītomic mass = (%1)(mass 1) + (%2)(mass 2) + ⋯Īnother example: oxygen exists as a mixture that is 99.759% 16O, 0.037% 17O and 0.204% 18O. The number with the decimals in it in the element box on the periodic table is the. This is repeated until there is a term for each isotope. (This is only possible because scientists. 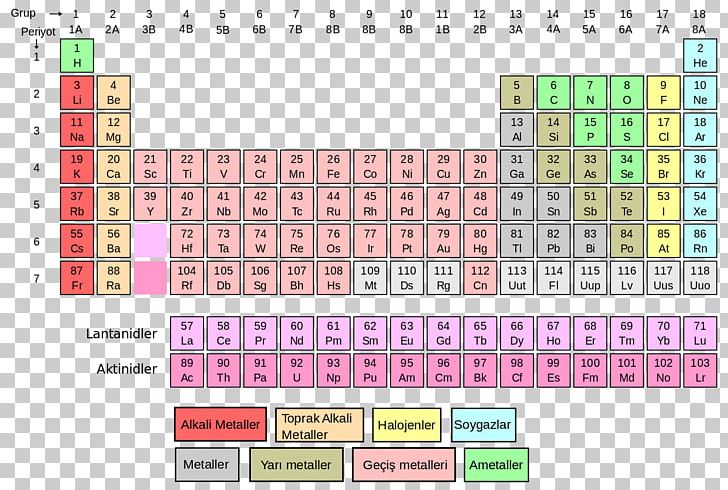
Atomic Mass Heat of Fusion Atomic Number Heat of Vaporization Atomic. Periodic Table 1 to 30 Element with Symbol and PDF without decimals- The sum of the masses of protons, neutrons, and electrons in an atom or group of atoms is called atomic mass. Fluorine: Density given for F at 0 Celsius. The weighted average is determined by multiplying the percent of natural abundance by the actual mass of the isotope. Atomic Mass of Elements 1 to 30 with Symbols PDF Download. 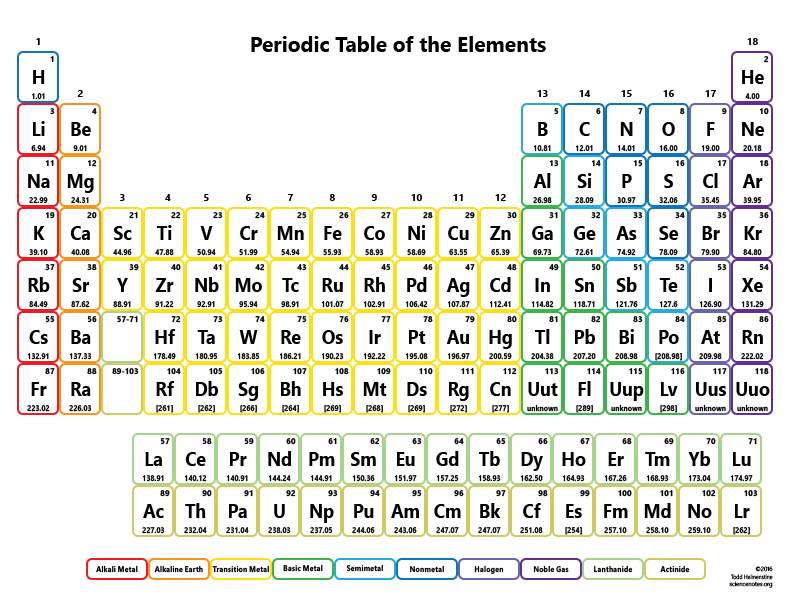
The atomic mass of an element is the weighted average of the atomic masses of the naturally occurring isotopes of that element. I also usually use the full number given on the periodic table just to be exact as well, but I have noticed some people round their masses from the periodic table to about 2 or 3 decimal places. Why? The reason is that we need to take into account the natural abundance percentages of each isotope in order to calculate what is called the weighted average. \( \newcommand = 35.968 \,u \nonumber \]Īs you can see, the average atomic mass given in the last column of the table above (35.453) is significantly lower. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
